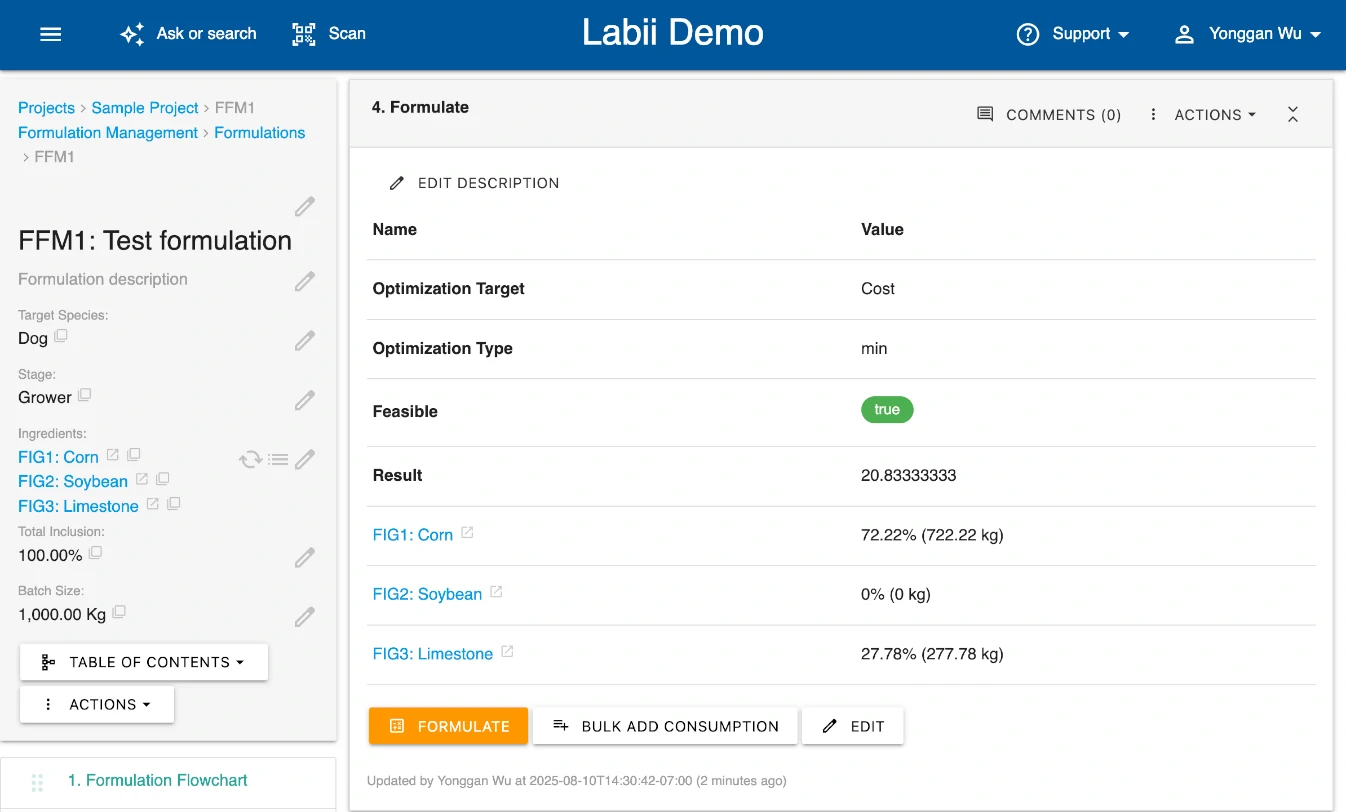
Design, optimize, and standardize formulations in one platform.

Design, optimize, and standardize formulations in one platform.
Apply constraints and optimization rules to reach feasible target formulations.
Scale formulations from R&D to manufacturing with full traceability.