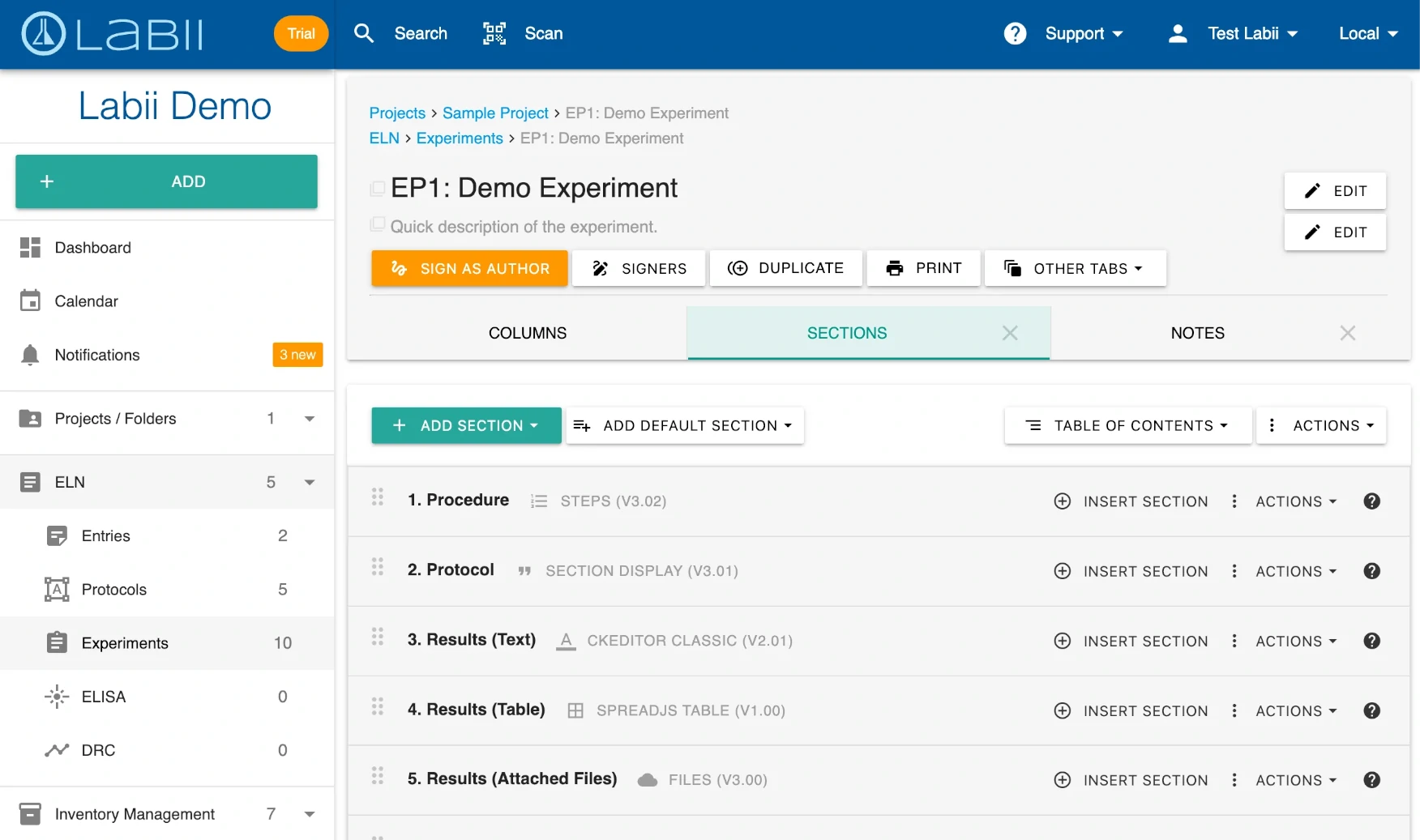
ELN for Production organizes experiments into structured sections for efficient, compliant documentation

ELN for Production organizes experiments into structured sections for efficient, compliant documentation
The experiment workflow guides users through each stage of production documentation